Clinical Trials Unit (BMEII-CTU)
The BioMedical Engineering and Imaging Institute Clinical Trials Unit (BMEII-CTU) is a modern hybrid between a contract research organization and an imaging core. We specialize in developing experimental imaging endpoints not previously used in multicenter clinical trials.
BMEII-CTU supports industry and federally sponsored multicenter clinical studies, as well as individual investigators interested in using imaging endpoints for their research. Typical services offered include trial design and consultation, multi-modality imaging protocol development, site training and qualification, image analysis, database management, data quality control and publication support. Qualified BMEII-CTU analysts are experienced in the use of FDA approved, 21CFR Part 11 compliant, image analysis platforms and adhere to ICH GCP regulations. Some examples of our work are:
Cardiovascular Imaging
BMEII has pioneered the non-invasive imaging of vascular disease using PET, CT and MRI to visualize anatomical and functional aspects of atherosclerosis and its sequelae. We image and assess the vasculature and other territories such as the airways, heart, spleen, bone marrow, brain and adipose tissue.
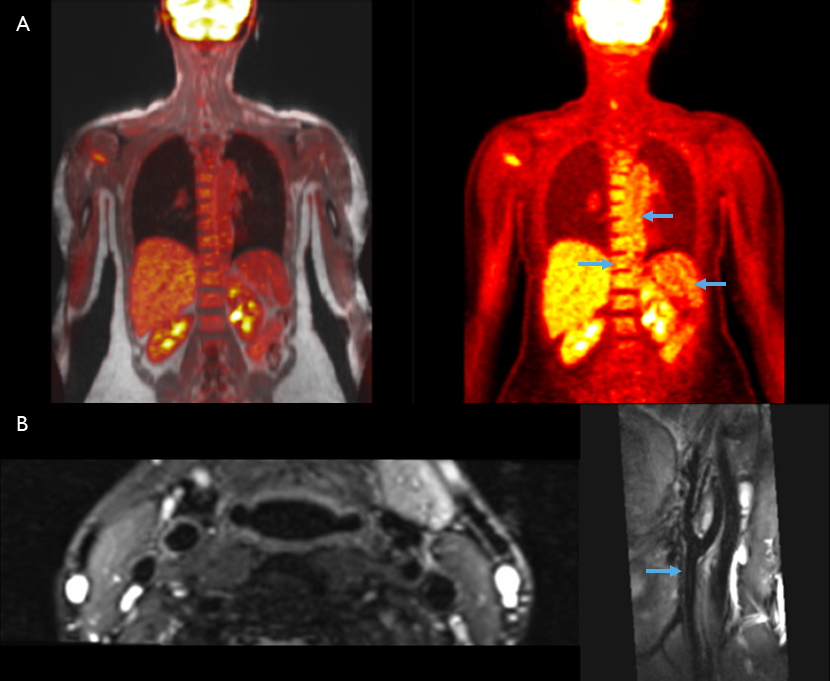
A) 18F-FDG-PET/MR and corresponding PET in the coronal plane showing uptake in the aortic wall, spleen and bone marrow.
B) High resolution, T2-weighted, black blood MR in the axial and parasagittal planes showing carotid artery walls.
Monitoring Disease Progression
Novel imaging endpoints can be used to evaluate the efficacy of a drug, or track disease progression, by imaging patients pre- and post-treatment. Quantitative and qualitative assessment of plaque components, and volumetric analysis of clot burden in the lungs and peripheral vasculature are some examples of imaging endpoints measured by BMEII-CTU for clinical trials.
3D volume rendered video of contrast enhanced CT Angiography showing diminishing clot burden after treatment in this patient with pulmonary emboli.
3D volume rendered video of contrast enhanced MR Venography showing diminishing clot burden after treatment in this patient with deep vein thrombosis.
Novel Application of Radiotracers
BMEII-CTU has expertise in the novel application of radiotracers. For example, 18F-NaF-PET is used to detect bone formation and remodeling. Uptake in plaque is believed to be related to vascular calcification.
Pseudoxanthoma elasticum (PXE) is an autosomal recessive disease that results in calcifications in the skin, eyes and cardiovascular system. Using 18F-NaF-PET in PXE patients can show active calcification, as identified in the bilateral femoral arteries in the video.
3D volume rendered video showing CT, 18F-NaF-PET, and fused PET/CT in a PXE patient.
Detection of Latent Disease
We can apply innovative imaging techniques for early detection of latent disease. For example, dual-energy CT (DECT) can distinguish different materials based on their relative absorption of X‐rays at different energy spectra.
DECT has been used to visualize gout in the joints, but our capabilities allow us to identify monosodium urate deposits in the vasculature. Quantifying these deposits could help assess treatment efficacy and correlate with future cardiovascular risk.
3D volume rendered video showing DECT of patient with gout. MSU deposits are depicted in green.
Faculty - Biomedical Engineering and Regenerative Medicine
The BioMedical Engineering and Imaging Institute (BMEII) at the Icahn School of Medicine at Mount Sinai (ISMMS), in partnership with Rensselaer Polytechnic Institute, is seeking mid to senior-level faculty members to spearhead a novel research initiative in the engineering of reparative and regenerative Medicine.
Faculty - Artificial Intelligence (AI) and Translation to Medicine
Faculty - Neuroengineering
Postdoctoral Fellow
The BioMedical Engineering and Imaging Institute at the Icahn School of Medicine at Mount Sinai is seeking a Postdoctoral Fellow.
News
Mount Sinai Researchers Featured in Nature for Advancing Healthspan Science
Nature has published a sponsored feature highlighting Mount Sinai’s leadership in redefining ageing through the XPRIZE Healthspan initiative. The article showcases the work of Drs. Miriam Merad, Zahi Fayad, and Fanny Elahi, who are pioneering new approaches to extend...
Dr. Zahi Fayad Featured in TIME for Innovative Research on Health Tracking and Longevity (Digital Twin Study)
“Let’s say you are stable, stable, stable, then suddenly start seeing a small dip in some measurements. That dip is presymptomatic, by the way ... That’s when I want to intervene.” — Zahi Fayad
[VIDEO] Dr. Zahi Fayad at Aspen Ideas 2025: Health Panel on Healthspan
What is health—and how do we measure it? Dr. Zahi Fayad emphasizes extending healthspan, which focuses on optimizing human health across the lifespan. By defining and tracking the trajectory of health, we can intervene earlier, and ensure people live not just longer,...
Latest Publications
Artificial intelligence–enabled rapid diagnosis of patients with COVID-19
Xueyan Mei, Hao-Chih Lee, […] Yang Yang
Nat Med (2020).
Probing myeloid cell dynamics in ischaemic heart disease by nanotracer hot-spot imaging
Max L. Senders, Anu E. Meerwaldt, ... Willem J. M. Mulder
Nat. Nanotechnol. 15, 398–405 (2020).